Carbon atoms in graphite12/16/2023  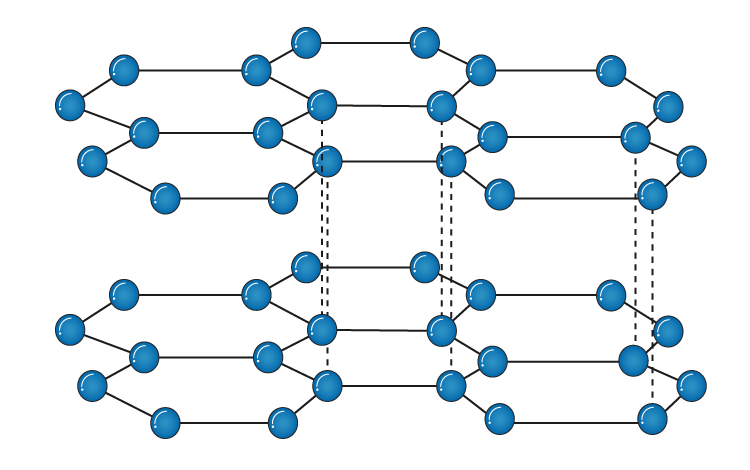
Each carbon atom joins four other carbon atoms in regular tetrahedrons (triangular. Each carbon atom has four valence electrons available to participate in the formation of chemical bonds. At ambient conditions, the stable bonding configuration of carbon is graphite. But in a graphite crystal, each carbon atom is joined to three other carbon atoms by covalent bonds to form hexagonal rings. The two most common allotropes of pure carbon are diamond and graphite. However, the latter two sets of angles are made up of three atoms in three different segments, so I wonder how or why they could be defined in "PRES 011C". This produces hexagonal rings of carbon atoms. prepared by vapor deposition of carbon atoms on highly-oriented pyrolytic. University of Michigan Library Publication date. silicates/toppar/top_silicates.inp, when you defined "PRES 011C", you used not only the data of "Angles along XY"(Boldface), but also those of "Angles along XX and XY" and "Angles along YY and XY". Diffusion of carbon atoms in natural graphite crystals Publisher. silicates/code/patches.dat, belowing lines were used to construct the topology file,īut in the file. When I read the codes, I found something I could not well understand. Graphite Stir Rod Stick Crucible Stir Rod Long Carbon Stirring Rod Graphite Crucible Stir Stick for Melting Casting Refining Gold. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
